New York, USA, March 09, 2026 (GLOBE NEWSWIRE) -- Herpes Zoster Market Poised for Robust Growth During the Forecast Period (2026–2036) Driven by Rising Incidence and Vaccine Adoption | DelveInsight
The herpes zoster market is experiencing steady growth, driven by the rising global incidence of shingles, particularly among aging populations. Increasing awareness of the disease burden and complications, such as postherpetic neuralgia, is boosting vaccine uptake. Additionally, the launch of emerging vaccines such as Amezosvatein (CRV-101) by Curevo, JCXH-105 by Immorna Biotherapeutics, and others will further boost the market growth.
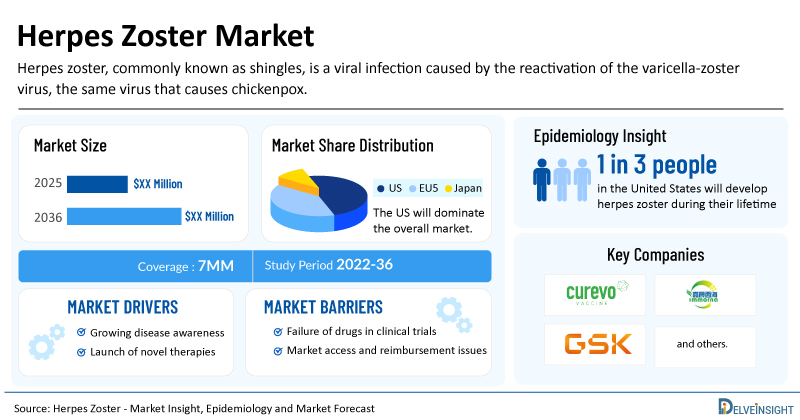
DelveInsight’s Herpes Zoster Market Insights report includes a comprehensive understanding of current treatment practices, emerging herpes zoster drugs, market share of individual therapies, and current and forecasted herpes zoster market size from 2022 to 2036, segmented into leading markets (the US, EU4, UK, and Japan).
Herpes Zoster Market Summary
- The total herpes zoster treatment market size is expected to grow positively by 2036 in the leading markets.
- The United States accounts for the largest market size of herpes zoster, in comparison to EU4 (Germany, Italy, France, and Spain), the UK, and Japan.
- About 1 in 3 people in the United States will develop herpes zoster during their lifetime. Most people have only one episode, but herpes zoster can recur.
- Key herpes zoster companies, including Curevo, Immorna Biotherapeutics, and others, are actively working on innovative herpes zoster drugs.
- Some of the key herpes zoster therapies in clinical trials include Amezosvatein (CRV-101), JCXH-105, and others. These novel herpes zoster therapies are anticipated to enter the herpes zoster market in the forecast period and are expected to change the market.
Discover the herpes zoster market size forecast 2026–2036 @ https://www.delveinsight.com/sample-request/herpes-zoster-market?utm_source=globenewswire&utm_medium=pressrelease&utm_campaign=spr
Ramandeep Singh, Senior Consultant of Forecasting and Analytics at DelveInsight, commented that herpes zoster poses a significant public health challenge due to its high incidence among older adults and the potential for severe pain and long-term complications. The introduction and widespread use of effective vaccines like SHINGRIX represent a major advancement in reducing the burden of this disease. Efforts should focus on improving vaccine uptake through public awareness campaigns and ensuring healthcare providers actively recommend vaccination to eligible patients.
Key Factors Driving the Growth of the Herpes Zoster Market
- Rising Incidence and Disease Awareness: Greater recognition of shingles incidence and its complications, such as postherpetic neuralgia (PHN), is driving demand. As awareness campaigns grow, diagnosis and preventive measures are adopted more widely.
- Aging Global Population: The risk of herpes zoster increases significantly with age, particularly among adults aged 50 and above, creating a growing target population for vaccination and treatment. Rising global life expectancy is rapidly expanding this age group.
- SHINGRIX Sets a New Standard in Shingles Protection: SHINGRIX, the licensed vaccine for herpes zoster, marks a major step forward in preventing the disease, providing more than 90% effectiveness even among older adults, who are at the highest risk for varicella-zoster virus reactivation.
- Launch of Emerging Herpes Zoster Vaccines: The dynamics of the herpes zoster market are expected to change in the coming years with the launch of vaccines such as Amezosvatein (CRV-101) by Curevo, JCXH-105 by Immorna Biotherapeutics, and others.
Herpes Zoster Market Analysis
- Treatment for herpes zoster must be initiated by a dermatologist or other healthcare provider within 3 days of rash onset.
- Ideally, if the treatment is initiated within three days, it can prevent possible complications, such as long-lasting nerve pain.
- Antiviral medications and vaccinations are often the most effective treatments for shingles, according to the National Foundation for Infectious Diseases.
- For shingles, SHINGRIX is available, while ZOSTAVAX, introduced in 2006, is no longer available in the US as of November 2020.
- The herpes zoster vaccine pipeline includes promising candidates such as Amezosvatein (CRV-101) by Curevo, JCXH-105 by Immorna Biotherapeutics, and others.
- They are currently being investigated in Phase II of clinical development and are expected to launch during the forecast period of 2026-2036.
Learn more about the best shingles vaccines approved @ Herpes Zoster Treatment Market
Herpes Zoster Competitive Landscape
Some of the herpes zoster vaccines in development include Amezosvatein (CRV-101) by Curevo, JCXH-105 by Immorna Biotherapeutics, and others.
Curevo’s CRV-101 is an experimental adjuvanted subunit vaccine being developed to prevent shingles in older adults. It was engineered to elicit a strong immune response while using a lower dose of adjuvant, aiming to match the effectiveness of the approved shingles vaccine while reducing side effects.
In March 2025, Curevo Vaccine announced it had secured USD 110 million in Series B funding to advance amezosvatein, its shingles vaccine candidate. In January 2025, the company reported positive updated safety and immunogenicity data from a 876-participant Phase II study of amezosvatein (CRV-101), in which the vaccine was directly compared with SHINGRIX in adults aged 50 and older.
Immorna Biotherapeutics’ JCXH-105 is a self-replicating RNA (srRNA) shingles vaccine. Unlike traditional mRNA platforms, srRNA generates temporary double-stranded RNA intermediates that boost innate immunity and strengthen T-cell–mediated adaptive responses, key for defending against herpes zoster. After intramuscular injection, JCXH-105 supports extended in vivo production of a modified VZV antigen, which may provide longer-lasting immunity and protection at lower doses than non-replicating mRNA vaccines.
JCXH-105 is currently in Phase II trials assessing its safety and immune response in healthy adults aged 50 and above. In May 2023, Immorna reported dosing the first participant in its First-In-Human, multi-center Phase I study of JCXH-105 for shingles prevention.
The anticipated launch of these emerging therapies are poised to transform the herpes zoster market landscape in the coming years. As these cutting-edge therapies continue to mature and gain regulatory approval, they are expected to reshape the herpes zoster market landscape, offering new standards of care and unlocking opportunities for medical innovation and economic growth.
To know more about emerging treatments for shingles, visit @ Herpes Zoster Medication
What is Herpes Zoster?
Herpes zoster, commonly known as shingles, is a viral infection caused by the reactivation of the varicella-zoster virus, the same virus that causes chickenpox. After a person recovers from chickenpox, the virus remains dormant in nerve tissues and can reactivate years later, often due to aging, stress, or a weakened immune system. This reactivation causes a painful, blistering skin rash that typically appears on one side of the body along a nerve pathway. In addition to the rash, individuals may experience burning, tingling, or shooting nerve pain. While shingles typically resolves within a few weeks, some people develop long-lasting nerve pain called postherpetic neuralgia. Vaccination can help reduce the risk and severity of herpes zoster.
Herpes Zoster Epidemiology Segmentation
The herpes zoster epidemiology section provides insights into the historical and current herpes zoster patient pool and forecasted trends for the leading markets. The lifetime risk of zoster is estimated to be at least 32%. Increasing age and cellular immunosuppression are the most important risk factors.
The herpes zoster market report proffers epidemiological analysis for the study period 2022–2036 in the leading markets segmented into:
- Total Incident Cases of Herpes Zoster
- Gender-specific Incident Cases of Herpes Zoster
- Age-specific Incident Cases of Herpes Zoster
- Eligible Patient Population for Treatment/Vaccination of Herpes Zoster
Download the report to understand herpes zoster 7MM epidemiology data @ Herpes Zoster Incidence
| Herpes Zoster Market Report Metrics | Details |
| Study Period | 2022–2036 |
| Herpes Zoster Market Report Coverage | 7MM [The United States, the EU-4 (Germany, France, Italy, and Spain), the United Kingdom, and Japan] |
| Herpes Zoster Epidemiology Segmentation | Total Incident Cases of Herpes Zoster, Gender-specific Incident Cases of Herpes Zoster, Age-specific Incident Cases of Herpes Zoster, and Eligible Patient Population for Treatment/Vaccination of Herpes Zoster |
| Key Herpes Zoster Companies | Curevo, Immorna Biotherapeutics, GlaxoSmithKline, and others |
| Key Herpes Zoster Therapies | Amezosvatein (CRV-101), JCXH-105, SHINGRIX, and others |
Scope of the Herpes Zoster Market Report
- Herpes Zoster Therapeutic Assessment: Herpes Zoster current marketed and emerging therapies
- Herpes Zoster Market Dynamics: Conjoint Analysis of Emerging Herpes Zoster Drugs
- Competitive Intelligence Analysis: SWOT analysis and Market entry strategies
- Herpes Zoster Market Unmet Needs, KOL’s views, Analyst’s views, Herpes Zoster Market Access and Reimbursement
Discover more about the competitive landscape for shingles treatment @ Herpes Zoster Clinical Trials
Table of Contents
| 1 | Herpes Zoster Market Key Insights |
| 2 | Herpes Zoster Market Report Introduction |
| 3 | Herpes Zoster Market Overview at a Glance |
| 3.1 | Market Share (%) Distribution of Herpes Zoster in 2025 |
| 3.2 | Market Share (%) Distribution of Herpes Zoster in 2036 |
| 4 | Executive Summary of Herpes Zoster |
| 5 | Key Events |
| 6 | Epidemiology and Market Methodology |
| 7 | Disease Background and Overview |
| 7.1 | Introduction |
| 7.2 | Signs and Symptoms |
| 7.3 | Risk Factors Associated with Herpes Zoster |
| 7.4 | Etiology |
| 7.5 | Pathogenesis |
| 7.6 | Diagnosis |
| 7.7 | Treatment |
| 7.8 | Prevention |
| 8 | Epidemiology and Patient Population |
| 8.1 | Key Findings |
| 8.2 | Assumptions and Rationale |
| 8.3 | Total Eligible Patient Population for Herpes Zoster Vaccination in the 7MM |
| 8.4 | The US |
| 8.4.1 | Total Incident Cases of Herpes Zoster in the US |
| 8.4.2 | Gender-specific Incident Cases of Herpes Zoster in the US |
| 8.4.3 | Age-specific Incident Cases of Herpes Zoster in the US |
| 8.4.4 | Eligible Patient Population for Treatment/ Vaccination of Herpes Zoster in the US |
| 8.5 | EU4 and the UK |
| 8.6 | Japan |
| 9 | Herpes Zoster Patient Journey |
| 10 | Marketed Herpes Zoster Drugs |
| 10.1 | SHINGRIX: Glaxosmithkline |
| 10.1.1 | Product description |
| 10.1.2 | Regulatory milestone |
| 10.1.3 | Other development activities |
| 10.1.4 | Safety and efficacy |
| 10.1.5 | Analyst Views |
| To be continued in the Report… | |
| 11 | Emerging Herpes Zoster Therapies |
| 11.1 | Key Cross Competition |
| 11.2 | CRV 101: CUREVO Inc. |
| 11.2.1 | Product Description |
| 11.2.2 | Other development activities |
| 11.2.3 | Clinical trial information |
| 11.2.4 | Safety and efficacy |
| 11.2.5 | Analyst View |
| 11.3 | JCXH-105: Immorna Biotherapeutics |
| To be continued in the Report… | |
| 12 | Herpes Zoster Market: Seven Major Market Analysis |
| 12.1 | Key Findings: Herpes Zoster Market |
| 12.2 | Key Herpes Zoster Market Forecast Assumptions |
| 12.3 | Conjoint Analysis |
| 12.4 | Herpes Zoster Market Outlook |
| 12.5 | Market Size of Herpes Zoster in the 7MM |
| 12.6 | Market size of Herpes Zoster by Therapies in the 7MM |
| 12.7 | Market Size of Herpes Zoster in the US |
| 12.7.1 | Total Market Size of Herpes Zoster in the US |
| 12.7.2 | Market Size by Therapies in the US |
| 12.8 | Market Size of Herpes Zoster in EU4 and the UK |
| 12.9 | Japan Herpes Zoster Market Size |
| 13 | KOL Views on Herpes Zoster |
| 14 | Herpes Zoster Market SWOT Analysis |
| 15 | Herpes Zoster Market Unmet Needs |
| 16 | Reimbursement and Market Access |
| 17 | Bibliography |
| 18 | Herpes Zoster Market Report Methodology |
Related Reports
Herpes Zoster Epidemiology Forecast – 2034 report delivers an in-depth understanding of the disease, historical and forecasted herpes zoster epidemiology in the 7MM, i.e., the United States, EU4 (Germany, Spain, Italy, and France), the United Kingdom, and Japan.
Herpes Zoster Clinical Trial Analysis
Herpes Zoster Pipeline Insight – 2026 report provides comprehensive insights about the pipeline landscape, pipeline drug profiles, including clinical and non-clinical stage products, and the key herpes zoster companies including EyeGene, Shulov Innovative Science, Dynavax Technologies, Curevo Vaccine, Vaccitech, NanoViricides, Pfizer, CPL Biologicals, Vapogenix, EuBiologics, Enzolytics, NAL Pharma, Gene One Life Science, Akshaya Bio, Turn Therapeutics, AIM Vaccine, XBiotech, HilleVax, Inc., and others.
Herpes Simplex Market Insights, Epidemiology, and Market Forecast – 2034 report delivers an in-depth understanding of the disease, historical and forecasted epidemiology, as well as the market trends, market drivers, market barriers, and key herpes simplex companies, including Teva Pharmaceutical Industries Ltd., Sanofi, Carlsbad Tech, Novartis AG, Emcure Pharmaceuticals Ltd., Fresenius Kabi AG, Viatris, Inc., Apotex Inc., Glenmark Pharmaceuticals Inc., GSK plc, and others.
Genital Herpes Market Insights, Epidemiology, and Market Forecast – 2034 report delivers an in-depth understanding of the disease, historical and forecasted epidemiology, as well as the market trends, market drivers, market barriers, and key genital herpes companies, including AiCuris, Heidelberg ImmunoTherapeutics, BioNTec, and others.
Postherpetic Neuralgia Market Insights, Epidemiology, and Market Forecast – 2034 report deliver an in-depth understanding of the disease, historical and forecasted epidemiology, market share of the individual therapies, and key PHN companies including Lexicon Pharmaceuticals, Merz Therapeutics, and others.
About DelveInsight
DelveInsight is a leading Business Consultant and Market Research firm focused exclusively on life sciences. It supports pharma companies by providing comprehensive end-to-end solutions to improve their performance. Get hassle-free access to all the healthcare and pharma market research reports through our subscription-based platform PharmDelve.

Contact Us Shruti Thakur info@delveinsight.com +14699457679 www.delveinsight.com
